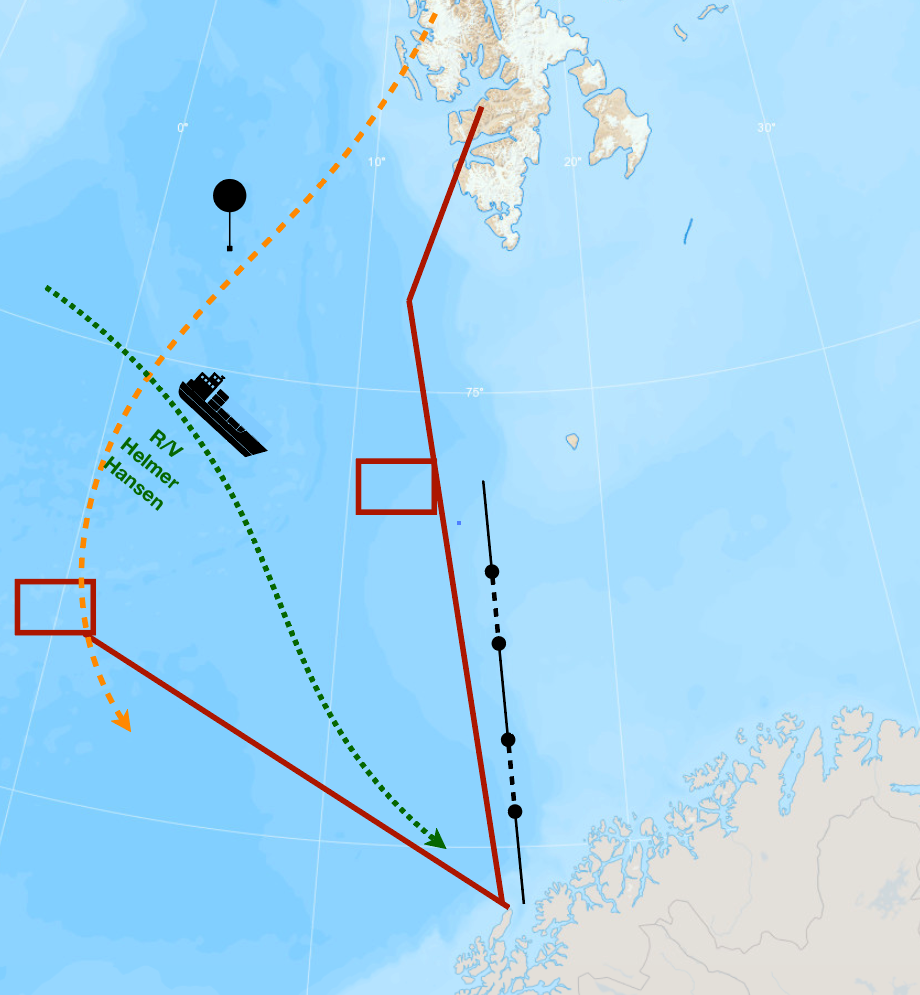
In a hangar in Kiruna in northern Sweden, three aircrafts are ready for take-off. Around 140 researchers, pilots and technicians have come to work with them. From Ny-Ålesund on Spitsbergen remotely operated balloons will drift southwards above the Norwegian Sea, where the research vessel Helmer Hanssen is heading toward the east coast of Greenland.
On Jan Mayen, Bear Island, Andenes and as far south as Ålesund, observers are ready to take water samples whenever it begins to rain or snow there. This will continue for three and a half weeks.
All this for a few drops of water. And yes, we are talking about plain, ordinary water.
An unusual field campaign
The unusualness lies in the intended role of this water. Measurements from its journey through the air will be used to distinguish the importance of evaporation and various cloud processes for rain and snow. With this information, scientists hope to find out how to program weather prediction models so as to produce the best forecasts of clouds and precipitation.
"Everything is almost ready now", says Harald Sodemann, professor at the Bjerknes Centre and the Geophysical Institute at the University of Bergen.
He leads one of the three research teams that will be present in Kiruna, responsible for one of the aircrafts. That three planes and their groups are there simultaneously, is a coincidence, but with a common goal of atmospheric exploration, the researchers can now cooperate.
Four days before departure, Harald Sodemann is still busy packing and organizing, but he has also had time to think about the weather.

Hopes for mildly bad weather
"The weather of my dreams is a cold air outbreak", says Harald Sodemann. "And we will get that."
He looks happily at a weather forecast chart on his monitor. The map shows a low-pressure center at the ice edge southwest of Spitsbergen.
During such conditions cold air from the ice-covered Arctic Ocean crosses the ice edge and flows southwards above open waters in the Greenland and Norwegian Seas – an outbreak of cold air from the ice over the ocean. The polar air is dry, and as it blows near the sea surface, it picks up huge amounts of water vapor from the sea. Strong winds enhance the evaporation, and the humid air is driven towards the coast of Norway.

When the air from the north and northwest hits land and rises against the mountains, heavy snow showers occur. This type of weather is associated with heavy snowfall in the coastal regions of northern Norway and with icing on ships. In extreme situations there is a risk of serious damage – by itself a good reason for research on weather like this.
In this case, there is another reason for selecting this kind of weather for a field campaign.
That the air streaming from the ice is cold, is not that important. What makes it interesting to Harald Sodemann and his team of researchers, is the amount of water vapor it contains:
Close to nothing.
"As the air above the ice is dry and remains so until it crosses the ice edge, we know that all the water it brings to Norway, originates from the ocean", he says.
In the event of a cold air outbreak, the researchers can follow the same water molecules all the way – from they evaporate from the ocean, while they condense into water droplets and freeze in the clouds, until they land in precipitation gauges or as snow crystals on the ground.
All they need to do, is catch them.
An airplane with a nose for clouds
"We will travel to places where water evaporates and then follow that water", says Harald Sodemann. "To do so, we must have a flying laboratory."
The airplane that will follow the journey of the water, belongs to the French National Centre for Scientific Research, CNRS. The vehicle is especially well constructed for flying inside clouds and is equipped with instruments that can register minuscular details in the clouds. Among other things, it can count cloud droplets, distinguish between water and ice and take frozen samples of water vapor from the air.
At any time, three team members will be occupied with weather forecasts. The airplane will be sent to locations where strong evaporation is predicted. Special models will be used to calculate how this air and its water vapor will move.
"If water evaporates at the ice edge and is predicted to be at Bear Island 24 hours later, we'll send the airplane to Bear Island", says Harald Sodemann. "After 48 hours the water vapor will be somewhere else", and we'll send the plane there."
The water will be traced.
Observers in Longyearbyen, on Bear Island, on Jan Mayen, on Andenes, in Abisko, in Kiruna and as far south as Ålesund will be ready to catch the water when it lands. If the weather forecast indicates that this exact water will fall as rain or snow somewhere between Andøya and Kiruna, the researchers will get in the car and drive there.
Remotely operated balloons
While aircrafts take off and land in Kiruna, six balloons will be sent up from the research station in Ny-Ålesund on Spitsbergen. The balloons will move up and down within the lower three kilometers above the ocean, remotely operated from the USA.
Along their southward-bound route they will secure data showing how moist air just above the sea mixes with drier air above – another process one must be aware of when trying to map the journey of the cloud water reaching the coast of Norway.
Aims to improve weather predictions
By studying how water circulates in nature, Harald Sodemann hopes to contribute to improving the representation of such processes in weather prediction models. That would allow for more accurate precipitation forecasts.
"Often, we encounter weather events when we cannot completely understand how a model reaches a specific result", he says.
A certain amount of rain can fall because a certain amount of water has evaporated from the ocean. But if the production of droplets from the vapor was more efficient, the same amount of precipitation requires less vapor. When limited to normal weather observations, one may not distinguish the importance of each factor.
To do that, the scientists need samples of the water and of the water vapor in the air. Ordinary water contains different isotopes – different versions of water molecules. These are heavy water and several forms of semi-heavy water, in addition to what we may call absolutely, ordinary water.
These isotopes all occur naturally in a drop of water. How much the drop contains of each isotope, depends on what the water has experienced during its journey.
As a result, Harald Sodemann and his researchers can use water samples to reconstruct the conditions in the ocean where the water evaporated, as well as what it has gone through while in the air and in the clouds. This way, they can separate the effects of different processes on the rain or snow that falls.
Important also for other weather types
The improvements they hope to contribute to in the models, will not be limited to cold air outbreaks. Their main reason for selecting such weather for measurements, is that the entire journey from the water takes off from the ocean until it lands on the ground, is completed in one to three days.
If they were to do anything similar for the low-pressure systems crossing the Atlantic towards Norway, they would have to follow the air over a much larger area for at least a week.
"We would have needed a lot of aircrafts", says Harald Sodemann.
Requesting assistance from Easter tourists
When aircrafts and researchers leave Kiruna right before Easter, some measurements still remain. The team hopes Easter ski tourists in northern Norway will help them take snow samples.
Will you go skiing in this region? Read about how you can take part in this citizen science project here.